
Why Is Lithium Used In Batteries: Today we can see small, powerful computers as small as to fit in our pockets easily such as a mobile phone. This is all because lithium-ion batteries can provide immense power at a very small size. It is due to lithium-ion batteries communications and transportation has advanced so much, which includes the shrinking of computers in size and the use of electric cars being practical.
These advancements were made possible by the fact that lithium-ion batteries could be more compact than the nickel-cadmium cells of the last generation while yet delivering the same amount of power. Even better, lithium-ion batteries use fewer harmful components and have a longer charge-retention time.
Lithium-Ion Battery Defined:
The lithium-ion battery belongs to the kind of batteries that can be recharged when used once. In this battery, lithium ions travel between the electrodes of battery via a conducting solution during charging and discharging. Graphite is a common material for cathode and a closely packed lithium compound for the positive electrode.
Why Is Lithium Used In Batteries
The energy density of lithium-ion batteries is very high, with a little memory effect and a significantly lower self-discharge rate. It is possible to construct cells that emphasize either power density or energy. However, because the solution present in these batteries can easily catch fire, these batteries can explode if damaged and pose a safety risk.
Lithium-ion Battery Working Principle:
The rocking chair concept governs how lithium-ion batteries function. Here, redox processes are responsible for transforming chemical energy into electrical energy. A lithium-ion battery is made up of two or more electrochemical cells that are electrically linked.
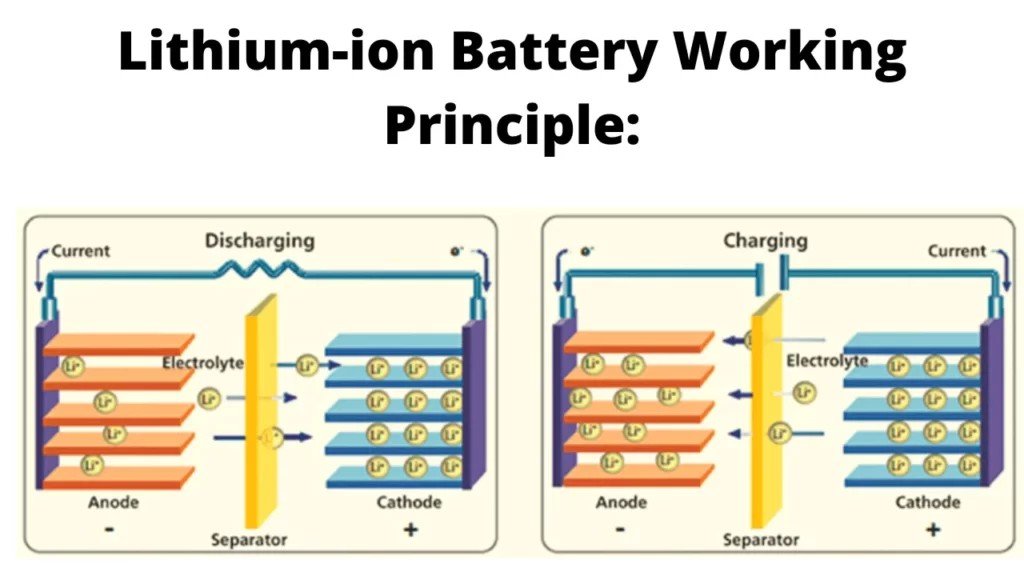
Ions travel toward the negative electrode, also known as the anode when the battery is fully charged. The lithium ions travel back to the top anode or cathode when the battery is fully depleted. This indicates that the lithium ions flow back and forth between the battery’s two electrodes during the charge-discharge operation.
Why Is Lithium The Best For Batteries?
Why is lithium the best for batteries? Lithium is considered the best for batteries because of several reasons. Lithium-based batteries are capable of providing more voltage per cell hence, reducing the number of cells required to achieve a certain voltage.
Due to this reason, the overall size of lithium battery is smaller compared to other battery technologies of same size.
This smaller size of Li-ion batteries have revolutionized industries all over the world. We can see computers getting smaller and smaller yet becoming more powerful. Also, electric vehicles make great use of Li-ion batteries because weight is the main component to handle when it comes to car. Lithium batteries deliver great power for electric vehicles yet keeping the weight in control.
Also, Lithium based batteries are considered to last longer than other rechargeable battery technologies. So, it is more convenient to use lithium based batteries in daily life consumer products. As, a result, we see great use of lithium batteries nowadays.
Uses of Lithium-ion Battery
Smartphones:
Lithium-ion batteries are used in almost all cellphones, including mobile phones, computers, wireless phones, etc. This excessive use is due to the portability, lightweight, and reusability of lithium-ion batteries. They are therefore ideal for portable devices. Additionally, these battery types offer a rapid charge rate, enabling the user to use their gadgets as needed with no disruption.
Cameras:
Because they can deliver a lot of power for a lot longer than conventional batteries, lithium-ion batteries are widely employed in DSLR cameras. Lithium-ion batteries are also chosen since they are lightweight and do not add bulk to the device. Some cameras also employ rechargeable lithium-ion batteries, which increase dependability and allow for several uses.
Power Banks:
A power bank is a small, portable device that enables users to quickly charge their smartphones, smartwatches, and other electronic gadgets wherever they are. A power bank commonly uses lithium ion-based batteries for this purpose. Lithium batteries are commonly used in power banks because of their small size and extraordinary life of lithium-based batteries.
Medical Equipment:
Lithium-ion batteries are used in various medical devices for diagnostic and therapeutic reasons, including hearing aids, surgical instruments, diagnostic equipment, cardioverter, cardiac pacemakers, healthcare professional robotics, injectables, and heart and vital signs sensors. Lithium-ion batteries have several benefits that make them ideal for medical devices, including their high reactivity, rapid charging time, lightweight, and non-hazardous nature.
Uninterrupted Power Supplies
In the event of a power outage or interruption, a UPS is a tool that offers temporary power backup. Lithium-ion batteries are frequently used in conjunction with such gadgets. This is due to the 50–80% reduction in floor area caused by the usage of battery cells in UPS equipment. Additionally, compared to a standard lead-acid-based UPS device, a lithium-ion battery-based Power supply weighs between 60 and 80 percent less.
Battery Operated Vehicles:
Lithium-ion batteries power various electric vehicles, including automobiles, motorcycles, scooters, and cycles. This is so because lithium-ion batteries outperform lead-acid batteries in terms of power-to-weight ratio, pressure and temperature endurance, and energy density. Additionally, they are safer, more efficient, and portable than conventional batteries.
Robotics
Lithium-ion batteries are widely favored in the research that deals with mechatronics. They power both industrial and humanistic robots.
Here is the Advantage And Disadvantages Of Lithium-ion battery
Lithium-Ion Battery Advantages
Using a battery with a Li-ion cell has several benefits. This is the reason for the vast application of lithium-based batteries in modern electronics. Anything, including mobile phones, computers, tiny electrical gadgets, automobiles, and several other uses. The advantages of lithium-ion batteries include:
1: Greater Energy Concentration:
Greater energy concentration is a great perk of lithium based batteries. High energy density battery tech is also required for electric cars. These lithium-ion batteries are also significantly better when we talk of power density. Energy density is important for batteries because computers and smartphones require a lot of power to carry complex calculations while still requiring great battery life.
2: Lesser Maintenance:
Low maintenance is another great perk of a lithium ion battery. Unlike lead acid batteries that require refilling of the electrolyte every once in a while, Li-ion batteries do not require any such maintenance. One of the benefits of lithium-based batteries is that it does not require to be regularly maintained. Ni-Cad batteries have to be periodically discharged to prevent the memory effect.
3: Lower Self-Discharging Rate:
The self-discharge rate of many batteries that can be recharged is a problem. The advantage of lithium-ion cells over other rechargeable cell types like Ni-Cad and NiMH is that their level of self-discharging is substantially lower. In the first four hours after being fully charged, it is usually around 5 percent, but it decreases to a rate of 1 or 2 percent every month.
4: Greater Voltage Per Cell:
Every lithium-based cell generates roughly 3.6 volts of electricity. A lithium-ion cell or battery exhibits respectable load characteristics. Before dropping off when the last charge is consumed, they offer 3.6 volts per cell very steadily. The output of every lithium-ion cell is more significant, needing fewer cells for many batteries uses. 3.6 volts per cells is quite impressive when compared to 1.3 volts provided by commonly used nickel based cells and 2 volts per cell provided by alkaline batteries.
Some cells require an additional process to be applied before their use after the first charge. At the same time, you can use lithium-based batteries directly after their first charge.
5: Multiple Types:
Lithium-ion batteries come in several different varieties. This benefit of lithium-based batteries may allow for the adoption of the appropriate technology for a particular application. Some li-ion batteries have high energy densities so are perfect for use in mobile electronics. Others can supply current levels that are significantly greater and are perfect for high power requiring tools and EV cars.
Lithium-Ion Battery Disadvantages
Any technology in this world has drawbacks to its use besides all the advantages Li-ion batteries also have some disadvantages. It is important to know the disadvantages of any component that you are using in your electrical components so that you can design your products keeping in mind the shortfalls of the component. The disadvantages of Lithium-ion battery include:
1: Requirement of a Battery Protective System:
The strength of lithium-ion batteries is not as high as that of specific rechargeable systems. They need to be guarded against being overcharged and discharged excessively. In addition, they must keep the current within acceptable limits. As a result, one drawback of lithium-ion batteries is that protective circuitry must be added to guarantee that they are kept inside their safe operating ranges.
Li-ion batteries may be utilized without technical understanding, thanks to the battery-saving circuitry. When the battery is fully charged, it may be left on charge, and the charger will switch off power to the battery. Lithium-ion batteries include built-in battery-saving systems that keep an eye on several elements of their performance.
2: Lifespan:
Lithium-based batteries degrade over time is one of the main drawbacks of using them in consumer products. A lithium cobalt oxide battery or cell for an average customer should be stored with a partial charge in a cold environment. Frequently, it takes about 500 to 1000 charge-discharge cycles for any rechargeable battery before their efficiency declines.
This depends on time or the date, but it also depends on how many charge-discharge cycles the battery has been through. The number of charge-discharge cycles are being increased as the technology advances but still if a battery is fitted into the device, there might be need for its replacement sometime.
3: Transportation:
Many airlines have a cap on the quantity of lithium-based batteries they may transfer, which means they can only do so via ships. Passengers have to carry li-ion batteries such as smartphones and power banks but in most cases there are restrictions on their transportation. Any lithium-based batteries that are transported separately must have covers, and other safeguards, to prevent short circuits.
4: Cost:
Costs of li-ion batteries are generally very high which is a major issue. Typically, the price of manufacturing a li-ion battery is forty percent more that the price to produce than nickel-cadmium cells. Cost of a product is very important when mass production of any technology is the purpose.
Developing Technology:
Even though lithium-based batteries have been used for quite some time, some people may still view them as being in the early stages of technical development. Because technology is constantly changing, this might be a drawback. However, the constant development of new lithium-ion batteries can also benefit as better options become available.
Daily Life Issues with Lithium-Based Batteries
Lithium-based batteries must be kept in a cold environment while not in use. However, this isn’t always a benefit or drawback. Lithium-ion battery aging is slowed as a result (and other chemistries). Experts advise keeping batteries at temperatures around 15 °C while they are still partially charged.
It is advised to partially charge and discharge lithium-based batteries so that a lesser number of charge-discharge cycles occurs and battery life can be increased. The ideal operating range for batteries has been suggested to be between 20 and 80 percent. Some battery-saving systems may prevent the cells from ever being fully charged or drained; this may already be accounted for in the amounts they report.
They do not want to discharge or fully charge lithium-ion batteries completely. This is because, in either condition, all of the lithium ions from one side will be eliminated, hastening the electrode’s decomposition.

